Foreign object infections: Difference between revisions
| Line 90: | Line 90: | ||
==Introduction== | ==Introduction== | ||
The dominant narrative in the literature for joint replacements and biofilms is that bacterial and fungal | The dominant narrative in the literature for joint replacements and biofilms is that bacterial and fungal (e.g. Candida Albicans) biofilms are responsible for chronic infections that lead to health problems. | ||
==Diagnostic testing== | ==Diagnostic testing== | ||
| Line 97: | Line 97: | ||
* Symptoms such as pain/redness/warmth/drainage at the incision site, swelling of the joint/limb, pain or stiffness in the joint after a pain-free period, chills or night sweats, and fatigue. | * Symptoms such as pain/redness/warmth/drainage at the incision site, swelling of the joint/limb, pain or stiffness in the joint after a pain-free period, chills or night sweats, and fatigue. | ||
* Blood tests to measure ESR (Erythrocyte sedimentation rate) and CRP (C-Reactive Protein). Both are associated with inflammation generated by infections. | * Blood tests to measure ESR (Erythrocyte sedimentation rate) and CRP (C-Reactive Protein). Both are associated with inflammation generated by infections. | ||
* Fluid samples from the joint. These samples can be cultured to look for bacteria and fungus/yeast. However, many potentially pathogenic microorganisms are difficult to culture so these tests may yield false negatives. Culture-negative PJI (Periprosthetic joint infection) has been reported in 7% of PJI episodes (see https://doi.org/10.1086/522184). | * Fluid samples from the joint. These samples can be cultured to look for bacteria and fungus/yeast. However, many potentially pathogenic microorganisms are difficult to culture so these tests may yield false negatives. Culture-negative PJI (Periprosthetic joint infection) has been reported in 7% of PJI episodes treated at Mayo Clinic Rochester from 1990-1999 (see https://doi.org/10.1086/522184). | ||
* Biopsy | * Biopsy | ||
| Line 117: | Line 117: | ||
==Treatment== | ==Treatment== | ||
Unfortunately, pathogens growing in biofilms are extremely difficult to eradicate with antibiotics | Unfortunately, pathogens growing in biofilms are extremely difficult to eradicate with antibiotics/antifungals and are resistant to eradication by the immune system. | ||
Removal of the medical device or prosthetic can eliminate the environment that helps biofilms thrive. However, certain explantation surgeries (e.g. pacemakers, heart valves) are very risky. | |||
Revision as of 15:56, 26 February 2022
Breast Implant Illness
Introduction
FDA data shows that chronic health problems are reduced in most patients who decide to have their breast implants removed (explantation). Nonetheless, breast implant illness (BII) is a controversial illness as many doctors do not believe that breast implants can lead to the development of chronic health problems. Despite presenting data on health outcomes following explantation, the FDA notes that "BII is not recognized as a formal medical diagnosis". BII occurs across all implant types, textured and untextured, as well as damaged and undamaged.
There is currently no test to detect BII and no test that can help predict the outcome of explantation. However, medical tests can find other reasons to explant such as a damaged implant.
One theory is that the bacterial biofilm colonies growing on breast implants are responsible for chronic health issues. A study by Lee at el. found that culturable bacteria and other microorganisms were 6 times more common in BII patients than non-BII patients with breast implants.
Overlap between BII and post-vax long haul
Both BII and vaccine injury show a significant overlap in symptoms. Whereas many vaccine injury and long COVID patients see a significant improvement in symptoms after several months, BII does not seem to exhibit a similar pattern. It is unclear if BII and long haul are two different facets of a similar process, or if they are distinct illnesses.
Because there is currently no test to differentiate between BII and long haul (aside from time), both patients and their doctors should consider the possibility of BII.
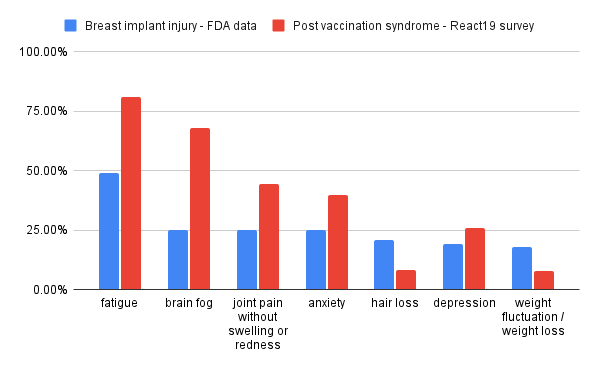
Symptom data from:
- https://www.fda.gov/medical-devices/breast-implants/medical-device-reports-systemic-symptoms-women-breast-implants
- https://www.react19.org/post/persistent-neurological-symptoms-patient-survey
FDA data
Outcomes
Of the 3,577 MDRs [Medical Device Reporting reports], 1535 (or 43%) reported the patient undergoing device explant at some time following the onset of symptoms. Of these, 1,473 reports provided information sufficient to calculate the time from implantation to explant. The average time to explant was 9 years, with a range of 0- 47 years. Only 290 MDRs provided information related to the status of a woman’s symptoms following explant. Of these, 279 noted improvement and 11 noted either no improvement or worsening of symptoms.
Most common systemic signs or symptoms
| Symptom | Percentage of MDRs (N=3,577) |
|---|---|
| Fatigue | 49% |
| "Brain Fog" | 25% |
| Joint Pain | 25% |
| Anxiety | 24% |
| Hair Loss | 21% |
| Depression | 19% |
| Rash | 18% |
| Autoimmune Diseases (incl. symptoms of autoimmune disease) | 18% |
| Inflammation | 18% |
| Weight Fluctuation | 18% |
Data on breast implant illness
- FDA data: https://www.fda.gov/medical-devices/breast-implants/medical-device-reports-systemic-symptoms-women-breast-implants
- NIH data: https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC8342259/
- 23% of patients reported complete resolution of symptoms following explantation, with 74% reporting partial resolution. Only 3% reported no improvement.
Scientific papers
- ‘ASIA’ – Autoimmune/inflammatory syndrome induced by adjuvants https://www.sciencedirect.com/science/article/abs/pii/S0896841110000788 - This review paper (over 800 citations) discusses how autoimmune syndromes are linked to vaccine adjuvants, breast implants, and Gulf war syndrome (GWS).
- Autoimmune dysautonomia in women with silicone breast implants https://doi.org/10.1016/j.jaut.2021.102631 - The authors discuss a correlation between auto-antibodies and symptom severity, suggesting that autoimmunity is the cause of symptoms. It is unclear if auto-antibody tests may be useful in diagnosing BII or predicting explantation outcomes.
- Breast Implant Illness: A Biofilm Hypothesis https://dx.doi.org/10.1097%2FGOX.0000000000002755
- Still's disease, lupus-like syndrome, and silicone breast implants. A case of 'ASIA'(Shoenfeld's syndrome) https://www.researchgate.net/profile/Miguel-Saavedra-13/publication/221738376_Still%27s_disease_lupus-like_syndrome_and_silicone_breast_implants_A_case_of_%27ASIA%27_Shoenfeld%27s_syndrome/links/0a85e5325f08a081a5000000/Stills-disease-lupus-like-syndrome-and-silicone-breast-implants-A-case-of-ASIA-Shoenfelds-syndrome.pdf
- Two hundreds cases of ASIA syndrome following silicone implants: a comparative study of 30 years and a review of current literature https://doi.org/10.1007/s12026-016-8821-y
- Chronic fatigue syndrome with autoantibodies—the result of an augmented adjuvant effect of hepatitis-B vaccine and silicone implant http://www.cfsitalia.it/Documenti/ChronicFatigue_HBV_vaccine_silicone_implants.pdf - This case report suggests that there are safety concerns with giving vaccines (specifically Hepatitis B) to individuals with silicone implants.
- Silicone Gel Breast Implants and Connective Tissue Disease—A Comprehensive Review https://sci-hub.se/https://doi.org/10.1080/08916931000148818 - Discusses the correlation between breast implants and scleroderma, a group of rare diseases that involve the hardening and tightening of the skin. Scleroderma is believed to be an autoimmune disease.
The scientific literature refers to BII as siliconosis, ASIA / autoimmune/inflammatory syndrome induced by adjuvants, and silicone implant incompatibility syndrome. Many articles on this topic can be found by searching Google Scholar.
Support groups
Support groups and communities can be found via Facebook and Youtube (search Youtube for "breast implant illness"). The largest Facebook support group is Breast Implant Illness and Healing by Nicole with 159k members.
They may share information on how to find doctors who are knowledgeable about BII.
Support groups may have certain biases such:
- The viewpoint that the capsule scar tissue is also a cause of BII and should be removed during explantation.
- Favoring explantation.
More resources
Breast Implant Illness .com has a wealth of information on symptoms, tests, surgeons, etc.
Introduction
The dominant narrative in the literature for joint replacements and biofilms is that bacterial and fungal (e.g. Candida Albicans) biofilms are responsible for chronic infections that lead to health problems.
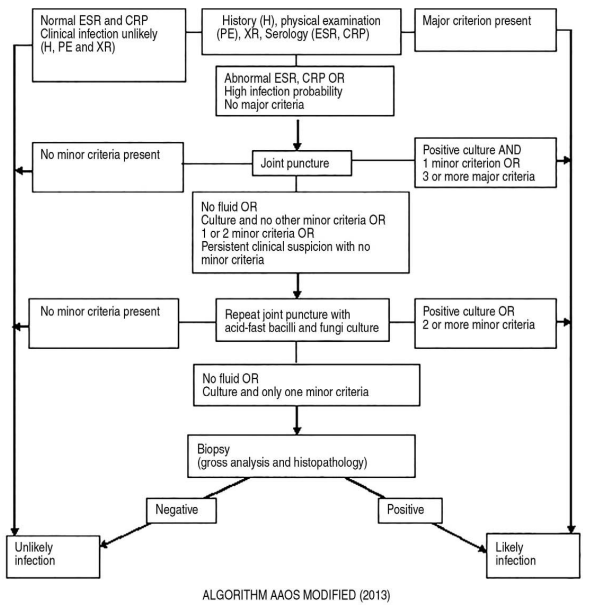
Diagnostic testing
The flowchart below shows one algorithm for diagnosing a periprosthetic joint infection (periprosthetic = close to prosthetic). Testing includes:
- Symptoms such as pain/redness/warmth/drainage at the incision site, swelling of the joint/limb, pain or stiffness in the joint after a pain-free period, chills or night sweats, and fatigue.
- Blood tests to measure ESR (Erythrocyte sedimentation rate) and CRP (C-Reactive Protein). Both are associated with inflammation generated by infections.
- Fluid samples from the joint. These samples can be cultured to look for bacteria and fungus/yeast. However, many potentially pathogenic microorganisms are difficult to culture so these tests may yield false negatives. Culture-negative PJI (Periprosthetic joint infection) has been reported in 7% of PJI episodes treated at Mayo Clinic Rochester from 1990-1999 (see https://doi.org/10.1086/522184).
- Biopsy
Source: Figure 1 from https://dx.doi.org/10.5371%2Fhp.2021.33.1.11
Other tests include:
- Elevated white blood cell count in synovial fluid (the fluid that lubricates joints).
- Elevated neutrophil percentage in synovial fluid.
- Purulence (pus) in the affected joint.
- Nuclear imaging
References:
- Current Guideline for Diagnosis of Periprosthetic Joint Infection: A Review Article https://dx.doi.org/10.5371%2Fhp.2021.33.1.11
- Periprosthetic joint infection: Current concept https://dx.doi.org/10.4103%2F0019-5413.106884
Treatment
Unfortunately, pathogens growing in biofilms are extremely difficult to eradicate with antibiotics/antifungals and are resistant to eradication by the immune system.
Removal of the medical device or prosthetic can eliminate the environment that helps biofilms thrive. However, certain explantation surgeries (e.g. pacemakers, heart valves) are very risky.