Enisamium iodide
Names: Enisamium iodide, Amizon / Amazon (trade name), carbabenzpiride
Recent studies have shown that the antiviral effect of enisamium is due to inhibition of viral RNA polymerase of influenza virus. See Wikipedia for references. The antiviral activity of enisamium iodide is associated with inhibition of influenza virus RNA polymerase. Enisamium iodide effectively inhibited SARS-CoV-2 virus replication in vitro in Caco-2 cells.
Clinical efficacy and safety
In 2010, the third phase of clinical trials on the basis of FBDU "Research Institute of Influenza" of the Ministry of Health of the Russian Federation (St. Petersburg, Russia) was completed with 100 patients (60 patients with influenza and SARS received enisamium iodide, 40 – placebo).
Machine translation of information on Amizon
From https://compendium.com.ua/dec/271524/#toc-6
Storage
Active ingredient: amizon ® (enisamium iodide)
1 tablet contains 125 mg (0.125 g) or 250 mg (0.25 g) of amizone ® (enisamium iodide);
excipients: lactose monohydrate, microcrystalline cellulose, povidone, croscarmellose sodium, calcium stearate;
shell: OPADRY II Clear 85F19250 (polyethylene glycol, polysorbate 80, polyvinyl alcohol, talc).
Medicinal form
Coated tablets.
The main physical and chemical properties: round, biconvex, yellow or yellow-green tablets, covered with a shell. On the surface of core tablets, the presence of minor inclusions is allowed.
Pharmacotherapeutic group
Antiviral means for systemic use. Antiviral means of direct action.
ATX code J05A X17.
Pharmacological properties
Mechanism of action
Antiviral effect of enisamium is associated with inhibition of the RNA polymerase of the influenza virus. Enisemium iodide effectively inhibited SARS-CoV-2 virus replication in vitro in Caco-2 cells.
Pharmacodynamics
Enisemium has an antiviral effect against various strains of influenza A virus (H1N1, H3N2, H5N1, H7N9), influenza B virus, respiratory syncytial virus, as well as strains of alpha-coronavirus NL-63 and beta-coronavirus SARS-CoV-2 in vitro.
Enisemium iodide has demonstrated efficacy against influenza A and B strains in in vitro studies using differentiated normal human bronchial epithelial (NHBE) cells, human hepatocellular carcinoma (HepG2) cells, human rhabdomyosarcoma (RD) cells, human colorectal adenocarcinoma cells ( Caco-2). In ferrets as a representative animal model for influenza research: enisemium iodide reduced the shedding time of influenza virus from ferret nasal washes compared to a placebo control group.
Clinical effectiveness
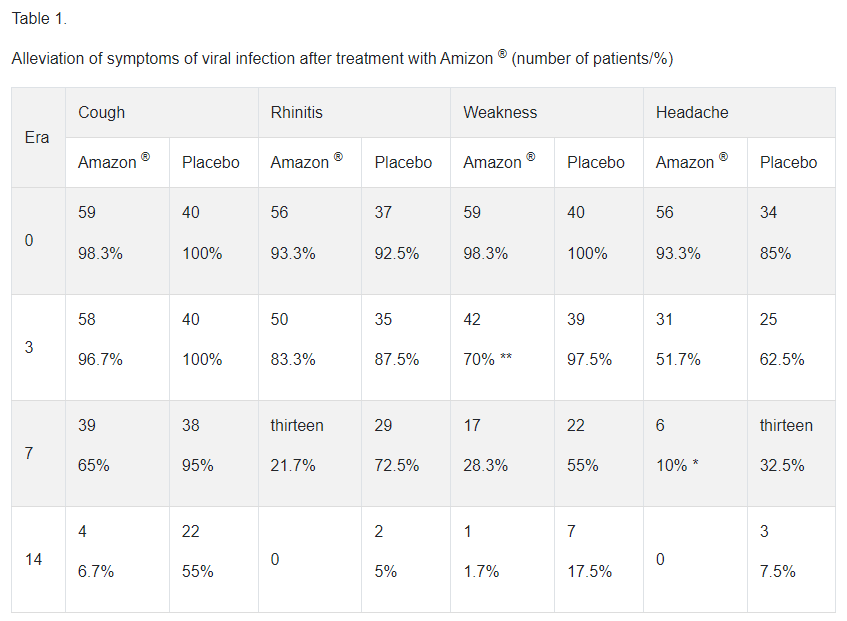
In a study in patients with acute respiratory viral infections, including influenza, treatment with enisemium iodide tablets in a daily dose of 1500 mg (2 tablets of 250 mg 3 times a day) provided positive dynamics of the disease, which was manifested by a more pronounced reduction in the symptoms of viral infection compared to with placebo (Table 1).
An early and statistically significant decrease in viral antigens in nasal swabs was found in patients who received Amizon ® compared to placebo (Table 2).
Enisemium treatment resulted in increased serum interferon levels compared to the placebo group.
The results of the third phase clinical study showed that enisamium iodide is well tolerated and clinically effective, which was demonstrated in the form of:
- reducing the duration of the period of elevated temperature by 1.1 days;
- reducing the duration of catarrhal and constitutional symptoms;
- reducing the use of expectorants and vasoconstrictors;
- reducing the number of days of incapacity for work;
- a reduction in the period of viral shedding and a significant reduction in the number of patients in whom viral antigens were detected, compared to the group of patients receiving placebo.
Greater effectiveness of enisanium was observed when treatment was started earlier.
Pharmacokinetics
Absorption
Enisemium iodide is quickly absorbed: the peak concentration in the plasma was reached 1.6–2.4 hours after a single dose. Absolute bioavailability in humans has not been studied, while relative bioavailability was less than 5%. The steady state in the case of oral administration of 500 mg three times a day and 1000 mg twice a day was reached after 3 days. Accumulation of the drug was not detected.
A study on dogs showed that when enisamium iodide is administered orally, 35% of the applied dose is absorbed from the gastrointestinal tract. Absorption in rodents was less than 5%.
Food markedly reduced the bioavailability of enisemium iodide. The mean values of C max and AUC inf when using 1500 mg after a meal were reduced by 46.8% and 26.6%, respectively, compared to such indicators when using 1500 mg on an empty stomach. The average value of t max increased after eating: when taken on an empty stomach — 0.75 hours, after eating — 2.75 hours.
Distribution
The degree of binding of enisemium iodide to serum proteins in humans is low.
Biotransformation
The starting compound (enisarium iodide) in humans is partially converted by hydroxylation, as well as binding to glucuronic acid (less than 5%). CYP 2D6 probably plays an insignificant role in the metabolism of enisemium iodide. Other studied cytochrome P450 enzymes do not have a significant effect on the processes of metabolic transformation of the original compound.
Elimination
Enisemium iodide is mainly excreted unchanged in the urine. After oral administration of radiolabeled enisium iodide to dogs, excretion in feces was 32–35%. The median half-life of single doses of enisanium iodide ranges from 2.69 to 3.35 hours and from 6.00 to 7.34 hours after repeated administration over 10 days.
Pharmacokinetics in separate groups of patients
Elderly patients
Pharmacokinetic studies involving elderly patients were not conducted.
Patients with liver and kidney damage
The pharmacokinetics of enisemium iodide in special populations have not been studied. However, based on the results of pharmacokinetic studies, taking into account the presence of renal and enteral excretion routes, as well as the low level of metabolism of enisium iodide, in subjects with organic damage to the liver and kidneys, significant accumulation of enisium in plasma should not be expected with short-term use (up to 7 days ) of this medicinal product. Thus, a worsening of the safety profile in patients with the above organic diseases is unlikely.
Clinical characteristics
Indication
Amizon ® is indicated for the treatment and prevention of influenza and acute respiratory viral infections.
Contraindication Hypersensitivity to drugs containing iodide, molecular iodine or covalently bound iodine, as well as to other components of the drug.
Interaction with other medicinal products and other types of interactions
Enisemium iodide can reduce the uptake of radioactive isotopes of iodine by the thyroid gland for up to 6 weeks.
The simultaneous use of iodine-containing drugs, as well as contrast agents and drugs containing covalently bound iodine should be avoided; treatment of wounds of a large area with the use of iodine-containing antiseptics (for example, molecular iodine) due to a possible increase in the risk of thyroid gland dysfunction.
Features of application
Taking enisamium iodide leads to an increase in the level of iodide in the plasma. A secondary increase in the level of circulating iodide triggers the mechanism of self-regulation of thyroid gland function, in which the capture of inorganic iodide by thyrocytes is inhibited, which helps to prevent the excessive formation of thyroid hormones; at the same time, the level of thyroid-stimulating hormone transiently increases (Wolf-Tchaikov phenomenon). This effect lasts for several days; after stopping the course of treatment, the function of the thyroid gland normalizes. In some cases, a transient increase in thyroid-stimulating hormone was observed for several weeks.
There is no information on the effect of enisanium iodide on patients with impaired thyroid function and patients who have previously developed hypothyroidism. However, it is advisable to monitor the function of the thyroid gland during treatment with enisemium iodide.
Other iodine-containing drugs are not recommended to be used during treatment and within 7 days after the end of treatment with enisamium iodide.
Excipients
Lactose content in Amizon ® 0.125 g tablet: 5.225 mg/table or 0.0153 mmol/table or 3.37% of the total weight of the tablet.
Lactose content in Amizon ® 0.250 g tablet: 10.45 mg/table or 0.0306 mmol/table or 3.37% of the total weight of the tablet.
If you have an intolerance to some sugars, consult your doctor before taking Amizon ® . Such patients can be prescribed capsules that do not contain lactose.
Sodium content in Amizon ® 0.125 g tablet: 0.285 mg/table or 0.0124 mmol/table or 0.18% of the total weight of the tablet.
Sodium content in Amizon ® 0.250 g tablet: 0.570 mg/table or 0.0248 mmol/table or 0.18% of the total weight of the tablet.
This medicinal product contains less than 1 mmol (23 mg) sodium/dose, i.e. essentially sodium-free.
Use during pregnancy or breastfeeding
It is contraindicated to use the drug during pregnancy, since clinical studies of enisamium iodide with the participation of pregnant women have not been conducted. Animal studies do not indicate direct or indirect effects on reproductive function/fertility.
Breast-feeding
It is not known whether enisemium iodide or its metabolites are excreted in human breast milk. It is not possible to exclude the risk of enisamium iodide entering the body of a newborn/infant.
The ability to influence the speed of reaction when driving vehicles or other mechanisms
Taking the drug Amizon ® does not affect the ability to drive vehicles or work with other mechanisms.
Method of application and dosage
Enisemium iodide tablets are swallowed without chewing 2 hours before meals.
The maximum single dose is 1000 mg (1 g), the maximum daily dose is 2000 mg (2 g).
Treatment. Enisemium iodide is prescribed to adults in a dose of 500 mg (0.5 g) 3 times or 1000 mg (1 g) 2 times a day.
Enisemium iodide is prescribed to children over 12 years of age in a dose of 500 mg (0.5 g) 3 times a day.
Children aged 6 to 12 years are prescribed 125 mg (0.125 g) 2-3 times a day.
The duration of treatment is 7 days.
Prevention . Adults and children over 16 years old — 250 mg (0.25 g) a day for 3–5 days, then — 250 mg (0.25 g) once every 2–3 days for 2–3 weeks; children aged 6–12 years — 125 mg (0.125 g) every other day for 2–3 weeks; children aged 12 to 16 years — 250 mg (0.25 g) every other day for 2–3 weeks.
Children
The drug in this medicinal form should not be used by children under 6 years of age.
Overdose
No reports of overdose with the drug Amizon ® have been received in clinical studies and during post-registration use.
There is no specific antidote.
Adverse reactions
The most common adverse reactions (ADRs) were taste disturbances, folliculitis, nasopharyngitis, headache, and lymphadenopathy (in placebo-controlled phase I studies). Most of these PRs were reported once and disappeared spontaneously. In most patients, these AEs did not lead to discontinuation of enisemium iodide.
Mild gastrointestinal disturbances (bitter taste in the mouth), heartburn, and burning in the throat were reported in a placebo-controlled phase III study.
Only adverse events that occurred more frequently in the enisemium iodide group than in the placebo group and that were reported in more than two subjects were considered.
Table 3 lists side effects observed during clinical trials and post-registration use of the drug. The frequency is defined as follows: very common (≥1/10), common (≥1/100, <1/10), uncommon (>1/1,000, <1/100) and frequency not known (cannot be determined from available data).
- Notification in the post-registration period.
Reporting of suspected adverse reactions
The reporting of suspected adverse reactions after the registration of a medicinal product is important. This ensures constant monitoring of the benefit/risk ratio of the use of the medicinal product. Healthcare professionals are asked to report all suspected adverse reactions through the state reporting system.
Expiration date
4 years.
Do not use the drug after the expiration date indicated on the package.
Storage conditions
Store in a place protected from light at a temperature not higher than 25 °C.
Keep out of the reach of children.
Packaging
Film-coated tablets, 0.125 g each. 10 tablets in a blister, 1 or 2 blisters in a pack or 20 tablets in a blister, 1 blister in a pack.
Film-coated tablets, 0.25 g each. 10 tablets in a blister, 1 blister in a pack or 20 tablets in a blister, 1 or 2 blisters in a pack.
Leave category
Without a prescription.
Producer
"Farmak" JSC.
The location of the manufacturer and its address of the place of business
Ukraine, 04080, Kyiv, st. Kyrylivska, 74.
Date of addition: 09/13/2022