Vaccine differences
Dosage
Moderna
- Adult dose: 100 micrograms
- Booster: 50 micrograms (source: the FDA states 0.25mL volume instead of 0.5mL)
Pfizer
- Adult dose and ages 12+: 30 micrograms
- Adult booster: 30 micrograms
- Ages 5-11: 10 micrograms: 10 micrograms (source: [FDA https://www.fda.gov/news-events/press-announcements/fda-authorizes-pfizer-biontech-covid-19-vaccine-emergency-use-children-5-through-11-years-age])
Spike protein
COVID vaccines all have differences in the spike protein sequence. The spike protein is made up of the S1 and S2 subunits joined at a furin cleavage site, where furin enzymes in the human host can cut the protein into two and enable the entry of the virus into human cells.
Two vaccines, Johnson&Johnson/Janssen and Novavax, use a modified furin cleavage site so that the spike protein is resistant against splitting/cleavage by furins. A letter from a Janssen representative briefly describes how the vaccine's modified furin cleavage site may cause it to be biologically different than a competing adenovirus-vector vaccine (Astrazeneca ChAdOx1 nCoV-19). A scientific paper published in Science discusses why the furin cleavage site was modified to increase vaccine effectiveness.
In theory, a modified furin cleavage site could prevent the spike protein from being split into S1 and S2. Full-length spike protein has different biological characteristics than the split subunits. However, in practice, vaccine injuries are occurring with all of the major vaccines being used (Sinovac, JnJ, AstraZeneca, Moderna, Pfizer). This may be due to all of the spike proteins ultimately being split and/or creating S1 subunits that are responsible for vaccine injury. See the etiology page for a discussion on the various theories regarding spike protein toxicity.
Some vaccines like Pfizer only contain the S1 spike protein (without S2), while other vaccines use "full length" spike protein that contains both subunits.
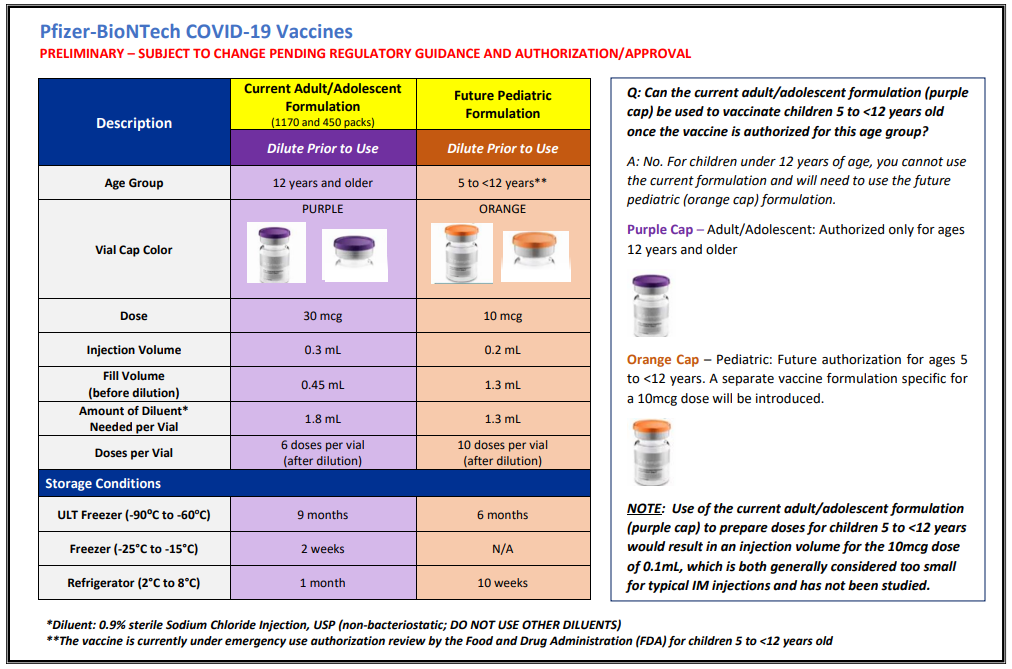
Adult versus pediatric formulation
A single page cheat sheet by the CDC summarizes differences between adult and pediatric formulations: https://www.cdc.gov/vaccines/covid-19/downloads/Pfizer-Pediatric-Reference-Planning.pdf
Due to the differences between the adult and pediatric formulations, the CDC recommends against using the adult/adolescent formulation in 5-11 year olds.