Enisamium iodide: Difference between revisions
(Created page with "'''Names''': Enisamium iodide, Amizon / Amazon (trade name), carbabenzpiride Recent studies have shown that the antiviral effect of enisamium is due to inhibition of viral RNA polymerase of influenza virus. See [https://en.wikipedia.org/wiki/Enisamium_iodide#cite_note-interfax.com.ua-5 Wikipedia] for references. The antiviral activity of enisamium iodide is associated with inhibition of influenza virus RNA polymerase. Enisamium iodide effectively inhibited SARS-CoV-2...") |
|||
| Line 43: | Line 43: | ||
Enisemium iodide has demonstrated efficacy against influenza A and B strains in in vitro studies using differentiated normal human bronchial epithelial (NHBE) cells, human hepatocellular carcinoma (HepG2) cells, human rhabdomyosarcoma (RD) cells, human colorectal adenocarcinoma cells ( Caco-2). In ferrets as a representative animal model for influenza research: enisemium iodide reduced the shedding time of influenza virus from ferret nasal washes compared to a placebo control group. | Enisemium iodide has demonstrated efficacy against influenza A and B strains in in vitro studies using differentiated normal human bronchial epithelial (NHBE) cells, human hepatocellular carcinoma (HepG2) cells, human rhabdomyosarcoma (RD) cells, human colorectal adenocarcinoma cells ( Caco-2). In ferrets as a representative animal model for influenza research: enisemium iodide reduced the shedding time of influenza virus from ferret nasal washes compared to a placebo control group. | ||
''Clinical effectiveness'' | |||
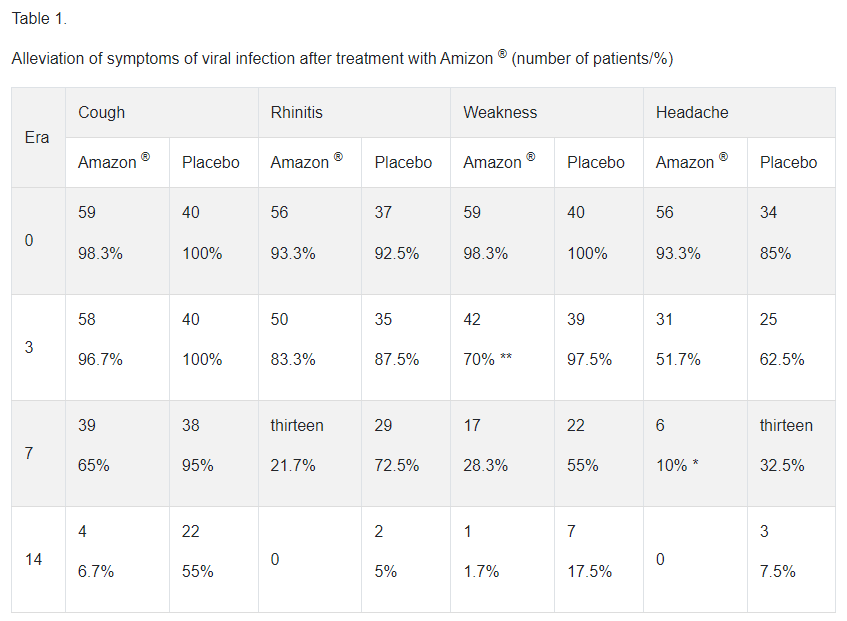
In a study in patients with acute respiratory viral infections, including influenza, treatment with enisemium iodide tablets in a daily dose of 1500 mg (2 tablets of 250 mg 3 times a day) provided positive dynamics of the disease, which was manifested by a more pronounced reduction in the symptoms of viral infection compared to with placebo (Table 1). | |||
An early and statistically significant decrease in viral antigens in nasal swabs was found in patients who received Amizon ® compared to placebo (Table 2). | |||
Enisemium treatment resulted in increased serum interferon levels compared to the placebo group. | |||
[[File:Amizon-table-1.png]] | |||
[[File:Amizon-table-2.png]] | |||
The results of the third phase clinical study showed that enisamium iodide is well tolerated and clinically effective, which was demonstrated in the form of: | |||
* reducing the duration of the period of elevated temperature by 1.1 days; | |||
* reducing the duration of catarrhal and constitutional symptoms; | |||
* reducing the use of expectorants and vasoconstrictors; | |||
* reducing the number of days of incapacity for work; | |||
* a reduction in the period of viral shedding and a significant reduction in the number of patients in whom viral antigens were detected, compared to the group of patients receiving placebo. | |||
Greater effectiveness of enisanium was observed when treatment was started earlier. | |||
''Pharmacokinetics'' | |||
''Absorption'' | |||
Enisemium iodide is quickly absorbed: the peak concentration in the plasma was reached 1.6–2.4 hours after a single dose. Absolute bioavailability in humans has not been studied, while relative bioavailability was less than 5%. The steady state in the case of oral administration of 500 mg three times a day and 1000 mg twice a day was reached after 3 days. Accumulation of the drug was not detected. | |||
A study on dogs showed that when enisamium iodide is administered orally, 35% of the applied dose is absorbed from the gastrointestinal tract. Absorption in rodents was less than 5%. | |||
Food markedly reduced the bioavailability of enisemium iodide. The mean values of C max and AUC inf when using 1500 mg after a meal were reduced by 46.8% and 26.6%, respectively, compared to such indicators when using 1500 mg on an empty stomach. The average value of t max increased after eating: when taken on an empty stomach — 0.75 hours, after eating — 2.75 hours. | |||
''Distribution'' | |||
The degree of binding of enisemium iodide to serum proteins in humans is low. | |||
''Biotransformation'' | |||
The starting compound (enisarium iodide) in humans is partially converted by hydroxylation, as well as binding to glucuronic acid (less than 5%). CYP 2D6 probably plays an insignificant role in the metabolism of enisemium iodide. Other studied cytochrome P450 enzymes do not have a significant effect on the processes of metabolic transformation of the original compound. | |||
''Elimination'' | |||
Enisemium iodide is mainly excreted unchanged in the urine. After oral administration of radiolabeled enisium iodide to dogs, excretion in feces was 32–35%. The median half-life of single doses of enisanium iodide ranges from 2.69 to 3.35 hours and from 6.00 to 7.34 hours after repeated administration over 10 days. | |||
''Pharmacokinetics in separate groups of patients'' | |||
''Elderly patients'' | |||
Pharmacokinetic studies involving elderly patients were not conducted. | |||
''Patients with liver and kidney damage'' | |||
The pharmacokinetics of enisemium iodide in special populations have not been studied. However, based on the results of pharmacokinetic studies, taking into account the presence of renal and enteral excretion routes, as well as the low level of metabolism of enisium iodide, in subjects with organic damage to the liver and kidneys, significant accumulation of enisium in plasma should not be expected with short-term use (up to 7 days ) of this medicinal product. Thus, a worsening of the safety profile in patients with the above organic diseases is unlikely. | |||
'''Clinical characteristics''' | |||
Revision as of 16:22, 13 October 2022
Names: Enisamium iodide, Amizon / Amazon (trade name), carbabenzpiride
Recent studies have shown that the antiviral effect of enisamium is due to inhibition of viral RNA polymerase of influenza virus. See Wikipedia for references. The antiviral activity of enisamium iodide is associated with inhibition of influenza virus RNA polymerase. Enisamium iodide effectively inhibited SARS-CoV-2 virus replication in vitro in Caco-2 cells.
Clinical efficacy and safety
In 2010, the third phase of clinical trials on the basis of FBDU "Research Institute of Influenza" of the Ministry of Health of the Russian Federation (St. Petersburg, Russia) was completed with 100 patients (60 patients with influenza and SARS received enisamium iodide, 40 – placebo).
Machine translation of information on Amizon
From https://compendium.com.ua/dec/271524/#toc-6
Storage
Active ingredient: amizon ® (enisamium iodide)
1 tablet contains 125 mg (0.125 g) or 250 mg (0.25 g) of amizone ® (enisamium iodide);
excipients: lactose monohydrate, microcrystalline cellulose, povidone, croscarmellose sodium, calcium stearate;
shell: OPADRY II Clear 85F19250 (polyethylene glycol, polysorbate 80, polyvinyl alcohol, talc).
Medicinal form
Coated tablets.
The main physical and chemical properties: round, biconvex, yellow or yellow-green tablets, covered with a shell. On the surface of core tablets, the presence of minor inclusions is allowed.
Pharmacotherapeutic group
Antiviral means for systemic use. Antiviral means of direct action.
ATX code J05A X17.
Pharmacological properties
Mechanism of action
Antiviral effect of enisamium is associated with inhibition of the RNA polymerase of the influenza virus. Enisemium iodide effectively inhibited SARS-CoV-2 virus replication in vitro in Caco-2 cells.
Pharmacodynamics
Enisemium has an antiviral effect against various strains of influenza A virus (H1N1, H3N2, H5N1, H7N9), influenza B virus, respiratory syncytial virus, as well as strains of alpha-coronavirus NL-63 and beta-coronavirus SARS-CoV-2 in vitro.
Enisemium iodide has demonstrated efficacy against influenza A and B strains in in vitro studies using differentiated normal human bronchial epithelial (NHBE) cells, human hepatocellular carcinoma (HepG2) cells, human rhabdomyosarcoma (RD) cells, human colorectal adenocarcinoma cells ( Caco-2). In ferrets as a representative animal model for influenza research: enisemium iodide reduced the shedding time of influenza virus from ferret nasal washes compared to a placebo control group.
Clinical effectiveness
In a study in patients with acute respiratory viral infections, including influenza, treatment with enisemium iodide tablets in a daily dose of 1500 mg (2 tablets of 250 mg 3 times a day) provided positive dynamics of the disease, which was manifested by a more pronounced reduction in the symptoms of viral infection compared to with placebo (Table 1).
An early and statistically significant decrease in viral antigens in nasal swabs was found in patients who received Amizon ® compared to placebo (Table 2).
Enisemium treatment resulted in increased serum interferon levels compared to the placebo group.
The results of the third phase clinical study showed that enisamium iodide is well tolerated and clinically effective, which was demonstrated in the form of:
- reducing the duration of the period of elevated temperature by 1.1 days;
- reducing the duration of catarrhal and constitutional symptoms;
- reducing the use of expectorants and vasoconstrictors;
- reducing the number of days of incapacity for work;
- a reduction in the period of viral shedding and a significant reduction in the number of patients in whom viral antigens were detected, compared to the group of patients receiving placebo.
Greater effectiveness of enisanium was observed when treatment was started earlier.
Pharmacokinetics
Absorption
Enisemium iodide is quickly absorbed: the peak concentration in the plasma was reached 1.6–2.4 hours after a single dose. Absolute bioavailability in humans has not been studied, while relative bioavailability was less than 5%. The steady state in the case of oral administration of 500 mg three times a day and 1000 mg twice a day was reached after 3 days. Accumulation of the drug was not detected.
A study on dogs showed that when enisamium iodide is administered orally, 35% of the applied dose is absorbed from the gastrointestinal tract. Absorption in rodents was less than 5%.
Food markedly reduced the bioavailability of enisemium iodide. The mean values of C max and AUC inf when using 1500 mg after a meal were reduced by 46.8% and 26.6%, respectively, compared to such indicators when using 1500 mg on an empty stomach. The average value of t max increased after eating: when taken on an empty stomach — 0.75 hours, after eating — 2.75 hours.
Distribution
The degree of binding of enisemium iodide to serum proteins in humans is low.
Biotransformation
The starting compound (enisarium iodide) in humans is partially converted by hydroxylation, as well as binding to glucuronic acid (less than 5%). CYP 2D6 probably plays an insignificant role in the metabolism of enisemium iodide. Other studied cytochrome P450 enzymes do not have a significant effect on the processes of metabolic transformation of the original compound.
Elimination
Enisemium iodide is mainly excreted unchanged in the urine. After oral administration of radiolabeled enisium iodide to dogs, excretion in feces was 32–35%. The median half-life of single doses of enisanium iodide ranges from 2.69 to 3.35 hours and from 6.00 to 7.34 hours after repeated administration over 10 days.
Pharmacokinetics in separate groups of patients
Elderly patients
Pharmacokinetic studies involving elderly patients were not conducted.
Patients with liver and kidney damage
The pharmacokinetics of enisemium iodide in special populations have not been studied. However, based on the results of pharmacokinetic studies, taking into account the presence of renal and enteral excretion routes, as well as the low level of metabolism of enisium iodide, in subjects with organic damage to the liver and kidneys, significant accumulation of enisium in plasma should not be expected with short-term use (up to 7 days ) of this medicinal product. Thus, a worsening of the safety profile in patients with the above organic diseases is unlikely.
Clinical characteristics